Will diamond replace silicon?
Following the advent of new renewable energy resources and the increased demand to convert and deliver such energy efficiently, there has been a remarkable increase in power electronics market research. Scientists and researchers are aiming to make our current technology smaller and faster.
However, they have run into several issues with material limitations directly related to power performance and efficiency. There has been a gradual shift from the augmentation of materials to replacing them as they become physically optimized.
Silicon has been the favored semiconductor material for over half a century now and still comprises nearly 95% of the market. Unfortunately, the advancement of silicon-based technology is limited by the physical nature of silicon. The materials and devices currently on the market are nearly perfect in terms of size and speed, leaving little room for improvement.
The more apparent problem that silicon-based electronics have is heat management. So silicon served the power electronics industry well but its days as go-to semiconductor material may be numbered.
The Properties that Make Diamond Shine
Diamond is a truly amazing material. It’s incredibly strong (due to its 3D structure and covalent bonds), near-transparent, and has high thermal conductivity. However, diamonds are also very good electrical insulators. This strangely makes it both useful and problematic for electrical devices.

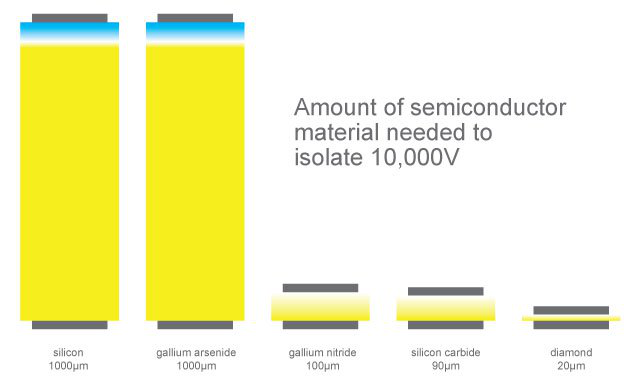
Table showing diamond properties against other silicon based materials.
This insulation ability allows for larger voltages and smaller leakage currents while the lack of conductivity means that electricity will not flow. However, the electrical conductivity can, in fact, be changed in an almost identical fashion to semiconductor doping.
Using elements such as phosphorus and boron, diamond can be doped with n-type and p-type materials which increase the electrical conductivity of diamond.
Diamond Semiconductors Research
The quest for silicon’s replacement as a semiconductor has been ongoing for years now. Recently, researchers have investigated tapping carbon nanotubes as a replacement for silicon or using insulating MOFs as an alternative semiconductor. Other teams are focusing their efforts on bypassing semiconducting material altogether with metamaterials that function like tiny valves.
However, some researchers also believe that diamond could be the looking glass in which the next semiconductor material lies
In comparison to its silicon counterpart, diamond can potentially exceed the relative efficiency of any silicon material property in regards to power electronics. Diamond can function as a wide-band gap semiconductor, has a high critical field temperature for superconducting, has a superior carrier mobility, and, above all, it has the greatest potential for heat conduction out of any accessible material.
The problem that scientists have had with using diamond is the fact that doping the crystals of diamond with boron calls for techniques that heat the diamond up to 1450 degrees Celsius. The issue with this was that the excess boron needed to be removed after the doping took place. Additionally, that technique only works effectively with diamonds that contain numerous crystals locked together into a single crystal. Diamonds that are composed of polycrystalline structures tend to have inconsistencies in the crystal structure that make them more difficult to work with.
Silicon helps diamond to replace silicon
A recent research paper was published in the Journal of Applied Physics describes a new method for doping diamond crystals using silicon, greatly increasing their potential as a semiconductor.
As described in their paper, a team comprised of University of Texas at Arlington and University of Wisconsin-Madison researchers has made a collective discovery in a new method for doping diamonds. The teams demonstrated a simple and efficient method for the selective doping of single crystalline diamonds (SCD) using thermal diffusion at significantly lower heat than that of current techniques.
The secret ingredient to the method turns out to be SILICON of all things!
The team discovered that if they bonded the SCD with silicon that had already been doped with boron and heated to 800 degrees Celsius, the boron would shift from the silicon to the diamond. This occurs due to the diamonds’ naturally shifting carbon, which leaves gaps for the boron atoms to fill.

The method described in their paper only addresses p-type semiconductors; a new method for doping n-type semiconductors is still a ways away.
And when it happens diamond can be a pioneer in the power electronics industry. Until then silicon will the king in the industry!
-Anirudh Kanfade